 We can write the configuration of oxygen's valence electrons as 2s2p. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell. Step-2: Need to do electron configuration of sodium. Valence electrons are the electrons in the outermost shell, or energy level, of an atom. Links the ion formation to the position of the element on the periodic table, and the valence. Group 1 elements have just one valence electron and group 18 elements have eight, except for helium, which has only two electrons total. First we need to know the total number of electrons in the. sodium loses an electron OR that fluorine gains an electron. In general, the number of valence electrons is the same within a column and increases from left to right within a row. Here is a table representing the number of valence electrons of elements belonging to the second period and their electronic configuration. How do you calculate the number of valence electrons in a sodium atom Step-1: Determining the total number of electrons in sodium. So, just by writing the electronic configuration of that element, we can quickly determine its number of valence electrons. The configuration of electrons gives a quick overview of the number of electrons present in the last shell. 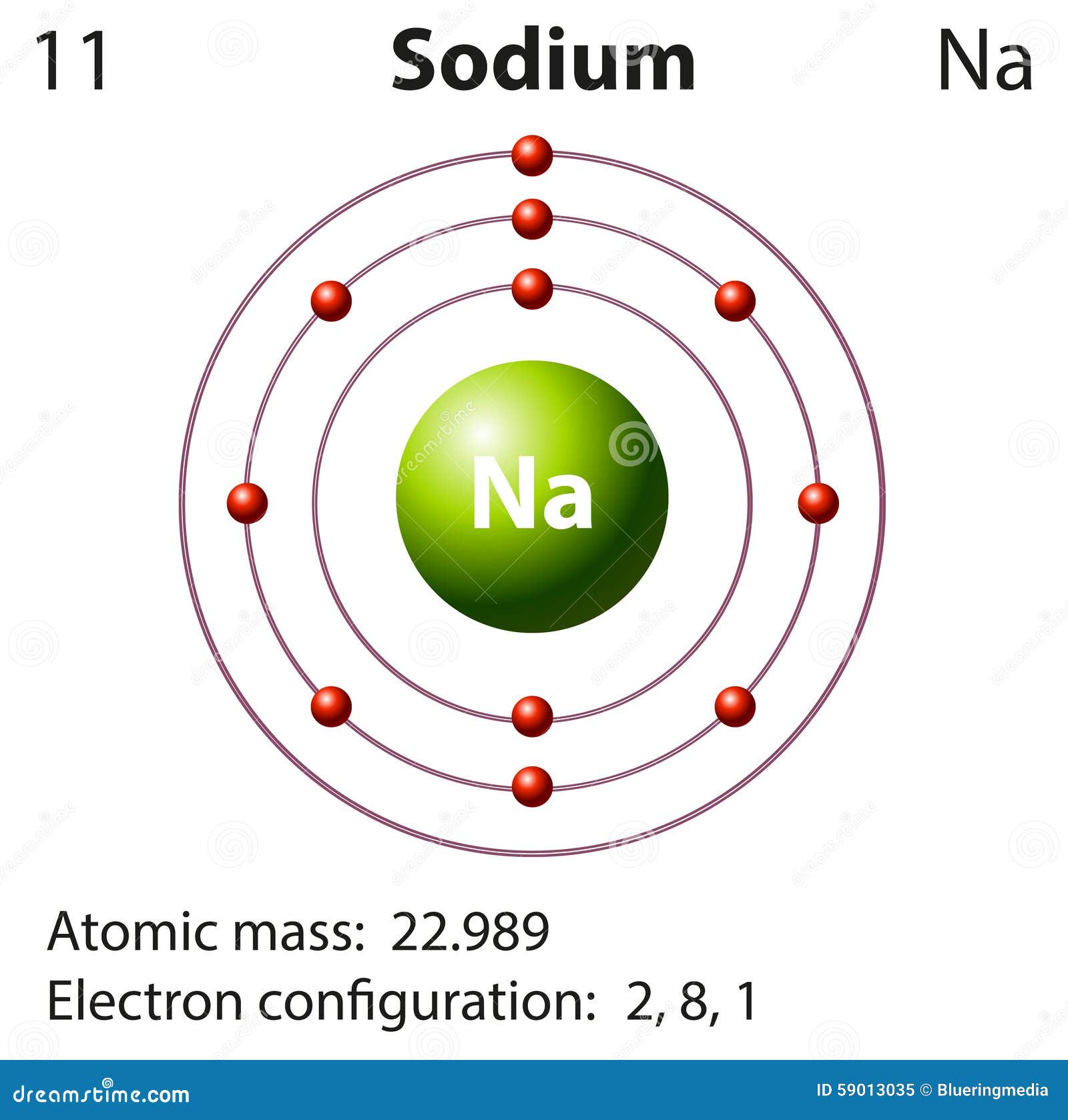
The arrangement of electrons in such orbitals is known as electronic configuration. Sodium has 1 valence electron because there is 1 electron present in the outermost shell of the Sodium (Na) atom. 
Complete step by step answer: - In the question it is asked to calculate the valence electrons present in the sodium atom. Within the shells, electrons occupy a special place called atomic orbitals. To know about the valence electrons present in an atom we should know the atomic number of the element. Identify a more reactive nonmetal element in the same period as Sulfur and Argon. Identify the element with 4 energy levels and 2 valence electrons. Identify the element with 7 valence electrons in period 4. It is a positive ion, because the number of protons (11+). Identify the element in period 5 and group 17. By Using Electronic Configuration of the Element Because the sodium atom has only one valence electron to lose, it tends to form only the 1+ ion. How can a sodium atom reach a full octet. The table below depicts the number of valence electrons in the different groups of the periodic table: Periodic Table GroupĢ. Sodium atoms have 11 total electrons in 2-8-1 configuration. Each atom not only sodium has valence electrons. It depends upon which period the element is in. The rule applies to the transition and inner transition elements in groups 3-12. Sodium (Na) has 11 electrons as its atomic number is also 11. However, this only holds for the main group elements, groups 1-2 and 13-18. Na has one valence electron since it is located in group 1 in the periodic table of elements and Cl has seven valence electrons. In contrast, the number of valence electrons across a period increases by one as we move left to right of a period.Įxception: As discussed, the period number indicates the number of shells, whereas the group number specifies the valence electron number in the outermost shell of an atom. As we proceed downwards in a group, the numbers of valence electrons are same, although the number of shells increases. Here, we just refer to the periodic table and search for the position of the element in it. On the other hand, fluorine has seven valence electrons, as shown. It is the most widely used method to determine the number of valence electrons in an element. All other elements in this family, Li, Na, K, Rb, and Cs, also have only one valence electron. 
All elements can be represented in this fashion.There are two ways of calculating the number of valence electrons in an element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
